Fibroids Quick Facts
Some Quick Facts about Uterine Artery Embolisation
Introduction
Uterine fibroid embolisation (UFE) (embolization) is being increasingly used in Europe and the USA for the treatment of fibroids. We have treated over 2,200 patients since December 1996. The procedure which is non surgical involves the occlusion of blood vessels supplying uterine fibroids. Uterine fibroids are benign growths of the uterine muscle occurring in 30-40% of women. Most fibroids do not cause any problems and do not require treatment. Some fibroids however can cause heavy periods which can lead to anaemia and debilitation, or if the fibroids grow large they can lead to 'compression syndrome' in which adjacent organs may be compressed such as the bladder leading to frequency of urination, the bowel leading to constipation and bloating or they may cause backache and sciatica.

Procedure
A tiny catheter is inserted under local anaesthetic into an artery in the right groin. Under x-ray control a micro catheter is introduced selectively into each of the two arteries that supply the uterus. The micro catheter is passed approximately half way down the artery and then fine particles of a solid substance called PVA (Poly Vinyl Alcohol) are injected through the catheter into the uterine artery. The particles are carried to the leash of vessels supplying the fibroids. These vessels become silted up thereby depriving the fibroid of blood which dies and shrinks.
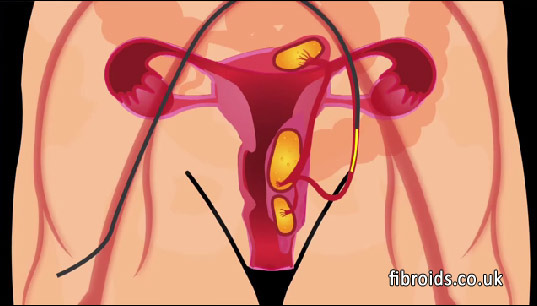
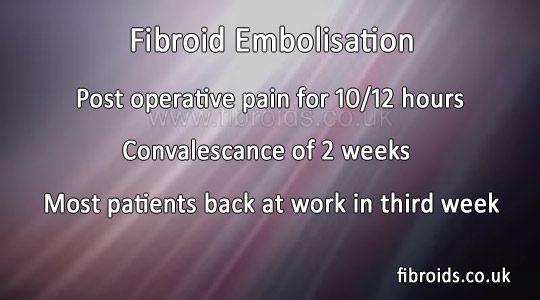
PVA is an inert harmless material which has been used to occlude vessels in other parts of the body for decades. (See video on website www.fibroids.co.uk) Following the procedure the patient usually experiences pain over the next 12 to 24 hours. The pain varies from mild to severe but is adequately controlled by analgesics. Occasionally over the next 3-4 weeks the patient may experience cramps and occasionally some bleeding and an mild intermittent temperature. Patients spend 2 days in hospital and are usually advised to take two weeks off work. In our series the average time to patients feeling completely 'normal' is 2.2 weeks. During the procedure intravenous sedation is administered.
World Experience
The world experience would indicate a very high success rate for fibroid embolisation. The main complication of the procedure is infection leading to hysterectomy. The incidence of this complication is less than 0.5% which is lower than the incidence of ovarian failure occurring following hysterectomy with conservation of the ovaries. A very small number of patients have stopped having periods altogether following the procedure. In our series we have not had a case of infection in our last 1,700 cases. None of the previous 5 cases were emergencies or required ICU admission.
Fertility (more detailed information)
In our trial of fibroid embolisation we have had 74 patients who have had successful completed pregnancies. Some of our patients had a history of infertility, som eof them had had previous myomectomies which had not resulted in pregnancy and some of our patients who had fibroid embolisation got pregnant after a subsequent myomectomy. The situation with regard to myomectomy versus embolisation for patiens who have fibroids and wish to become pregnant is comlex and patients need to be assessed on a case by case basis. A detailed examinaton of the issues surrounding fibroid embolisation and myomectomy in fertility patients cabe be found on our website fibroid.co.uk together with a review of the literature with references. Patients who are considering fibroid embolisation and who are wishing to become pregnant should carefully read this section of our website.
The main surgical alternative to embolisation for those wishing to retain their reproductive potential is myomectomy. You should ask your gynaecologist about the realistic chances of a successful myomectomy and the likelihood of this improving fertility in the case of your particular fibroid problem.
Current Experience
Dr W J Walker, Consultant Interventional Radiologis, has so far carried out fibroid embolisation on over 2,400 patients. He has been carrying out embolisations for conditions other than fibroids since 1974. Most patients have had a successful outcome with marked reduction or elimination of their symptoms and a reduction in fibroid size of between 47% and 100% - average 61%.
Complications
Mortality The mortality from UFE is almost negligible and is certainly less than 1 in 30-40,000 compared to a mortality for hysterectomy for fibroids of 1 in 1,000-1,500. Non fatal complications In the Guildford/London Clinic series of over 2,400 patients, 5 infective complications have occurred leading to hysterectomy. These were all in our first 500 cases, none were emergencies and none required ICU admission. Although the complication rate of fibroid embolisation is very low and significantly less than any surgical procedure, complications can occur - these are:
-
Infection leading to hysterectomy: This is very rare in our series there have been none in the last 1,700 patients and only 5 in the whole series of over 2,200 patients. There have been no emergency hysterectomies or intensive care unit admissions.
-
Ovarian failure: Under 45 this is rare with an incidence of less than ½%. Ovarian failure leads to a premature menopause and a patient over 45 would normally then take HRT which these days is very effective. It should be remembered that around 2 -3% of women will go into the menopause under 45 normally.
-
Vaginal discharge: This is usually transient and self limiting. Around 5-6% of women will have discharge necessitating hysteroscopy to remove the cause. Patients having fibroid embolisation in Guildford or at The London Clinic are part of an ethically approved observational trial. However, NICE has approved fibroid embolisation as an accepted treatment since 2006 and it is no longer necessary for it to be a trial procedure. Overall the complication rate of fibroid embolisation is significantly less than hysterectomy for fibroids which has a serious complication rate of greater than 4%.

Failures
Analysis of our early figures including long term 5 year follow up, indicates a failure rate for UFE of 8% and a success rate of 92%
Status of Uterine Fibroid Embolisation
Uterine fibroid embolisation is a Recommended Treatment for fibroids:
August 2008 Practice Recommendations from American College of Obstetricians and Gynecologists say uterine fibroid embolisation is ‘Safe and Effective’ based on good, consistent Level A Scientific Evidence.Uterine fibroid embolisation was passed by NICE (National Institute for Clinical Excellence) for routine use in 2007.
Uterine fibroid embolisation has been elevated to a Best Practice Tariff for Uterine fibroid embolisation by the NHS.
Please note
Patients who are taking drugs such as Zoladex or Synarel will need to stop these for at least 6 weeks before the embolisation procedure. We require a clear cervical smear test to have been carried out within 18 months prior to fibroid embolisation. We accept both insured and self funding patients: Fibroid embolisation is covered by most insurance companies (OPCS code: XR360). For patients insured with BUPA, Dr Walker's BUPA provider number is 01460483.
Please confirm with your
insurance provider that they will cover your consultation, ultrasound and MRI scans together with blood tests which are
required prior to the procedure. Please also confirm with your insurance provider that they will cover your follow up
consultations and scans (minimum of 2) after the procedure.
For more information regarding self funding prices at the London Clinic, Nuffield Health Guildford Hospital or any other
private clinic Dr Walker works at, please contact Dr Walker's Executive Assistant on 07795 643019 or via email:
wjwalker@uk-consultants.co.uk
© Copyright 2015 • All Rights Reserved | Dr. W.J. Walker |